Earlier this year, actor James Van Der Beek succumbed to an advanced colorectal cancer. The shock over his death came not from the diagnosis, but from his age—48 years old. We have become accustomed to the prevalence of cancerous tumors correlating roughly to increased age, but this sad news was a bellwether of a disturbing health crisis: the dramatic rise in colorectal cancer diagnoses in midlife.
Epidemiology of Colorectal Cancers
The American Cancer Society has tracked statistics involving colorectal cancer among Americans by age, and their findings were alarming. While colorectal cancer incidence and mortality among the older population (older than age 50) have declined since the beginning of this century, incidence and mortality among the young adult population (ages 20-49) dramatically moved in the opposite direction!1
Some oncologists project that by 2030, one-third of all colorectal cancers will be diagnosed in persons younger than 50 years of age. Screenings must begin now!
Why were there such diverse trends between those 2 age groups? More aggressive efforts at early detection and improved treatments in the older population—in whom we expect to see more tumors—are readily absorbed explanations. Conversely, the opposite trend in the young adult population puzzles many medical experts. The alarming rate of increased colorectal cancer in the young adult population is finally being publicly recognized although its presence is not new in the medical literature.2-4 Some oncologists now project that by 2030, one-third of all colorectal cancers will be diagnosed in persons younger than 50 years of age.4 Hypothesized explanations are numerous: processed foods, excess red meat, obesity, lack of exercise, low dietary fiber, poor rate of screening, etc.2 It will take years of epidemiological studies to sort out the answers, but early aggressive screenings must begin now!
A Personal Screening Conundrum
“Hi. I’m from Dr. X’s office. We have your Cologuard screening test results. It’s positive.”
“Oh,” I paused, “and what does that mean?”
“That’s all I know now. Dr. X will contact Dr. Y to schedule a colonoscopy.”
Those were not reassuring words. Uncertain of my medical status, I consulted the website of the provider of the screening kit and scrolled through the FAQs. Those referred me to a paper by Thomas Imperiale, MD,5 that clarified the issue: Although often presented as a screening test for colon cancer, it’s really an initial screen for colon abnormalities that could indicate a cancer. Dr. Imperiale reported on a population at average risk for colon cancer (approximately 10,000 participants) between the ages of 50 and 84; 16% of individuals screened by this method were positive—like me. Those with positive results were referred for immediate colonoscopies: 4% were diagnosed with colon cancer, 51% had colonic polyps, and the remainder were noncancerous cases, cause unknown.5 I felt better but was still somewhat miffed that a nonmedically trained individual gave me the news!
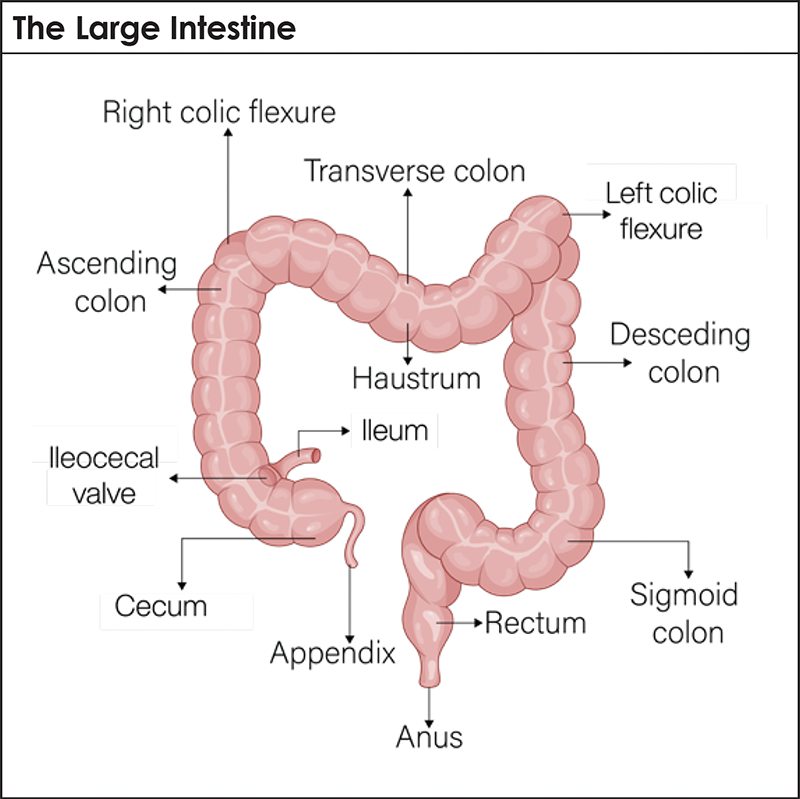
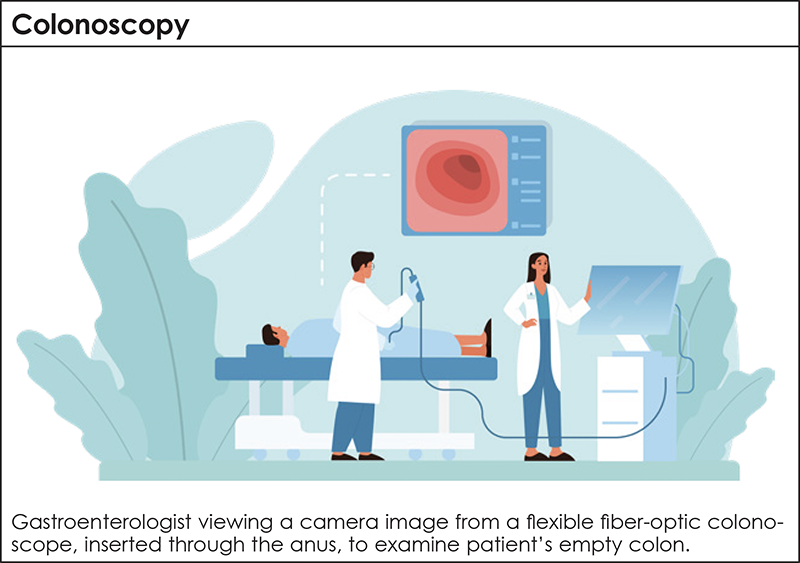
My subsequent colonoscopy by a veteran gastroenterologist went smoothly. He surgically removed 2 polyps from different areas of my colon and sent them off for a biopsy. Pathologists later confirmed this tissue as “not atypical.” I was in the clear.
In-Home Screening Kits: A Useful First Step
Unlike earlier immunochemically based stool tests like FIT or FOBT, a newer test analyzes stool DNA (sDNA).5 This test is commercially marketed as the in-home screening test Cologuard and is considered a breakthrough in the colorectal screening enterprise. The patients who use it, however, need to understand what it can and cannot do. The test does not definitively diagnose colorectal cancer but tells you whether you merit a colonoscopy, which can accurately diagnose such a cancer.
Cologuard5-8 analyzes sDNA biomarkers to monitor disorders within the colon. A next-generation test, Cologuard Plus, has been approved by the FDA and is currently undergoing clinical evaluation. Applying a new panel of molecular biomarkers, its early use indicates that, relative to the original Cologuard test, high sensitivity was maintained with a decline in the number of false positives that caused unnecessary colonoscopies.7-9 That’s a significant issue for elderly patients who tend to be at higher risk for complications from a colonoscopy.8
Those in the medical testing business often market their products to physicians using the clinical categories of sensitivity and specificity. Sensitivity measures the proportion that accurately identifies every case of the disease; 100% sensitivity refers to a test that has zero false negatives. Specificity measures the proportion that accurately identifies every case lacking the disease; 100% specificity refers to a test that has zero false positives. Realistically, it would be a very rare medical test that had both 100% sensitivity and 100% specificity. But the closer the test comes to that, the better for the patient. Cologuard reports the sensitivity and specificity for identifying colorectal cancer are determined as a result of both the kit’s usage and colonoscopy confirmation. Sensitivity of the assay was reported to be at least 92%, significantly higher than those earlier FIT or FOBT assays.5,7,8
Making Informed Decisions and Current Screening Recommendations
The American Cancer Society and the American Medical Association would be delighted if everyone aged 45 and older reported faithfully every 3 years for a colonoscopy. Of course, this is quite unrealistic. The inconveniences of colonoscopy make compliance an issue. Encumbrances include the dreaded “day of preparation” during which your colon is emptied using powerful laxatives; the cost of the procedure; sedation; support persons needed for transport, etc. The utility of an in-home noninvasive screening test that is mailed out for analysis fulfills a great need. But who is and who is not eligible for properly utilizing such a test?
Guidelines
Research6-8 shows that individuals who would not benefit from an in-home screening test include those who have…
- A history of colorectal cancer or any other cancer of the digestive system
- A family history of colorectal cancer
- A history of inflammatory bowel disease (IBD), Crohn’s disease, or ulcerative colitis
- Familial adenomatous polyposis (FAPa) or a family history of it
- Been diagnosed with Lynch syndromeb or have a family history of it
- Had a previous positive in-home screening test
- Had prior radiation therapy in the abdominal or pelvic areas
- Reached 75 years of age6 (deferred only at the discretion of their physician)
- aFAP is the presence of hereditary precancerous polyps proliferating in the colon. They very frequently progress into a cancer10 and can be surgically removed during a colonoscopy
- bHereditary genetic disorder that can cause nonpolyposis colorectal cancer (HNPCC)11
Included here is information for individuals who may benefit from an in-home screening test:
- If you are eligible as an average-risk patient, first see your healthcare provider who will facilitate ordering an in-home screening kit. (This article has highlighted the results of only 1 available commercial kit, Cologuard. Other tests are in the process of coming onto the clinical market. Consult your healthcare provider as to their availability and efficacy.)
- Among those eligible, everyone 45 years or older should be screened.1 Younger patients should be screened at the discretion of their physician
- Persons of African-American, Alaskan Native, or Eastern European Jewish descent should also inquire of their physician any special criteria for their screening11
- Medicare Part B and many private insurers will cover the cost of an in-home screening kit6,9
References
- American Cancer Society. Colorectal Cancer Facts & Figures. 2026. Accessed March 17, 2026. www.cancer.org/research/cancer-facts-statistics/colorectal-cancer-facts-figures.html
- Connell LC, Mota JM, Braghiroli MI, Hoff PM. The rising incidence of younger patients with colorectal cancer: questions about screening, biology, and treatment. Curr Treat Options Oncol. 2017;18:23.
- Teng A, Nelson DW, Dehal A, et al. Colon cancer as a subsequent malignant neoplasm in young adults. Cancer. 2019;125:3749-3754.
- Mauri G, Patelli G, Crisafulli G, et al. Tumor “age” in early-onset colorectal cancer. Cell. 2025;188:589-593.
- Imperiale T, Ransohoff D, Itzkowitz S, et al. Multitarget stool DNA testing for colorectal cancer screening. N Eng J Med. 2014;370:1287-1297.
- Dwyer A. What is the best way to screen for colorectal cancer? Conquer: the journey informed. February 2024, Accessed March 16, 2026. www.conquer-magazine.com/articles/what-is-the-best-way-to-screen-for-colorectal-cancer
- Exact Sciences Corporation. What is the Cologuard PlusTM test? Accessed March 17, 2026. www.cologuard.com/what-is-cologuard-plus
- Imperiale TF, Porter K, Zella J, et al. Next-generation multitarget stool DNA test for colorectal cancer screening. N Eng J Med. 2024;390:984-993.
- Schoenfeld P. Multi-target stool DNA test for CRC screening: how accurate is the new version? May 14, 2024. Accessed March 17, 2024. https://gi.org/journals-publications/schoenfeld_May2024/
- Johns Hopkins School of Medicine. Familial adenomatous polyposis. Accessed March 17, 2026. www.hopkinsmedicine.org/health/conditions-and-diseases/familial-adenomatous-polyposis
- Johns Hopkins School of Medicine. Colon cancer. Accessed March 17, 2026. www.hopkinsmedicine.org/health/conditions-and-diseases/colon-cancer
About the Author
Dr. Marley currently teaches within the faculty of the Department of Microbiology
& Molecular Genetics at Oklahoma State University. He holds a PhD in microbiology
and molecular biology from the University of Maryland (at Baltimore) and an
MSc degree in pathobiology from the Johns Hopkins Bloomberg School of Public
Health. In 2023, he was successfully treated for throat cancer at the Stillwater
Cancer Center in Oklahoma. He can be reached at